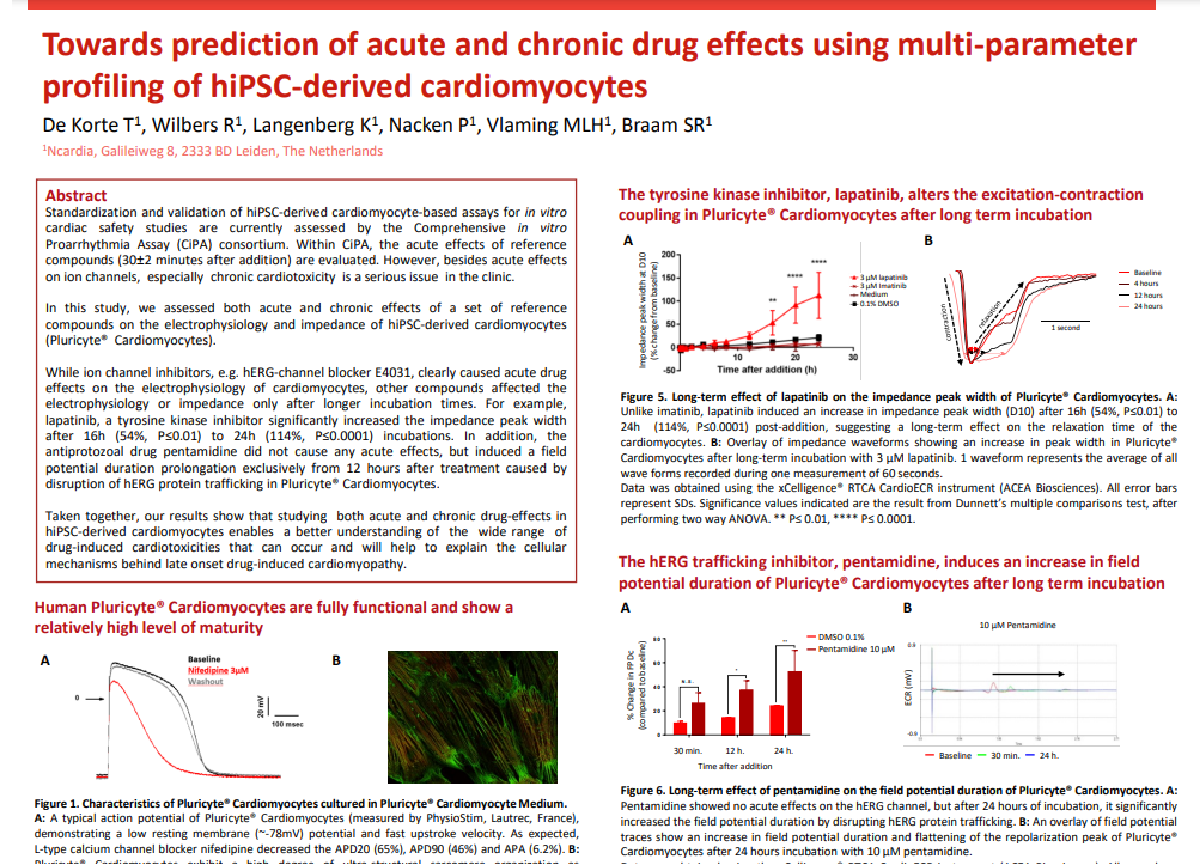
Contractility
Contractility is the force of contraction of cardiac and skeletal muscle cells, which can be disturbed in hypertrophic cardiomyopathy or muscular dystrophy, among other pathological conditions. This assay uses human-iPSC derived cardiac or skeletal muscle cells to mimic physiological tissue elasticity and applies electrical impedance to measure drug-induced effects on cell contractility and viability and it is compatible with medium throughput.
- Mimic physiological tissue elasticity
- Multiplexing options
- Non-invasive and label-free
Do you want to explore how this assay can help you progress your drug discovery programs?
Applications
Human iPSC-derived cardiac or skeletal muscle cells can be used to develop disease models with altered contractility. Our team can combine impedance analysis with multi-electrode array technology to simultaneously assess contractility, viability, and electrophysiology, providing a complete understanding of compounds’ efficacy, safety and toxicity at the early stages of drug development.
Simultaneous MEA and impedance analysis on human iPSC-derived cardiomyocytes for efficacy or safety assessments.
In this project we measured field potential duration and contractility in parallel, saving time and resources, and obtaining a more complete overview of compound effects. In this example we used isoproterenol, a compound known to affect the field potential and contractility by the activation of β-adrenergic receptors.
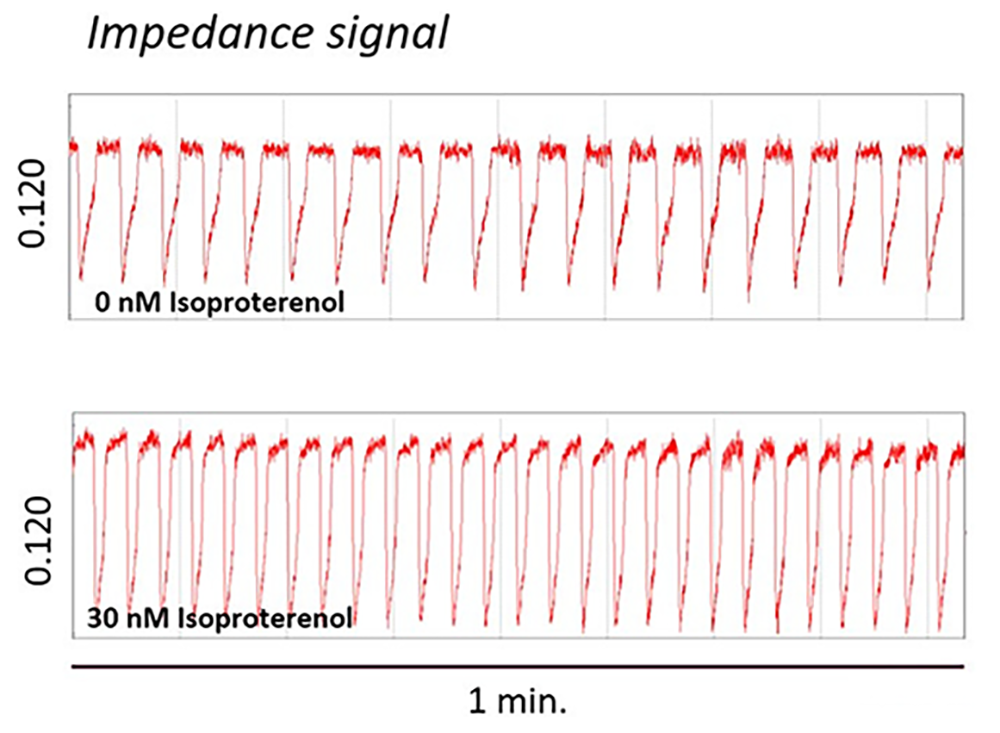 Treatment of human iPSC-cardiomyocytes with Isoproterenol increases the impedance peak amplitude and beat rate (measured by MEA) indicating a positive inotropic effect.
Treatment of human iPSC-cardiomyocytes with Isoproterenol increases the impedance peak amplitude and beat rate (measured by MEA) indicating a positive inotropic effect.
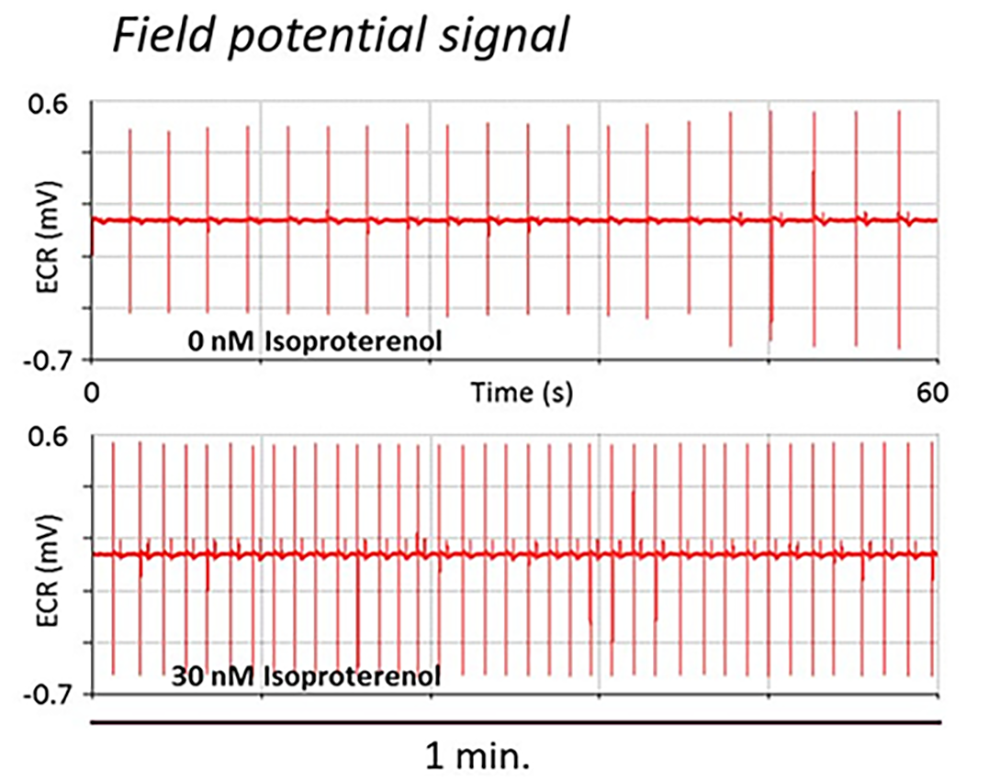 Treatment of human iPSC-cardiomyocytes with Isoproterenol increases the impedance peak amplitude and beat rate (measured by MEA) indicating a positive inotropic effect.
Treatment of human iPSC-cardiomyocytes with Isoproterenol increases the impedance peak amplitude and beat rate (measured by MEA) indicating a positive inotropic effect.
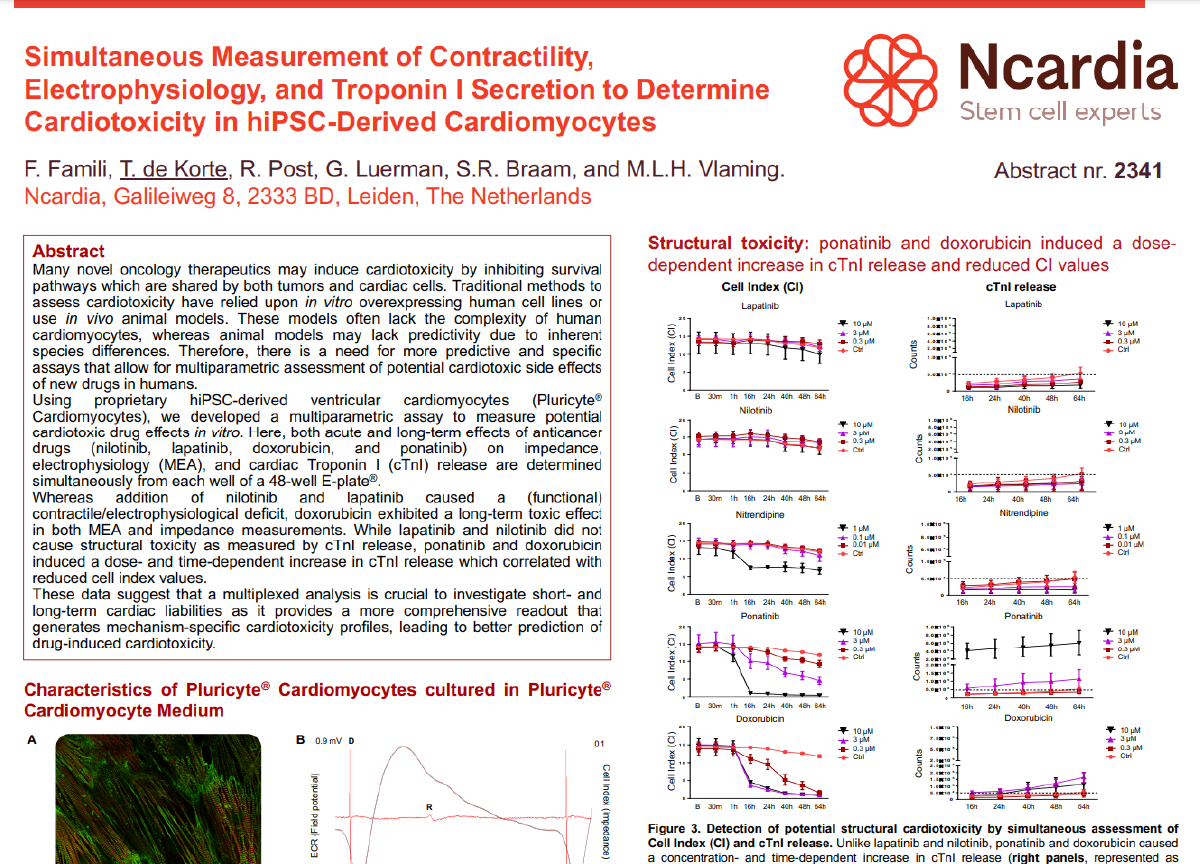
Evaluating acute and chronic cardiotoxic effect of oncology therapeutics on the contractility and viability of human iPSC-derived cardiomyocytes.
Many oncology therapeutics may induce cardiotoxicity by inhibiting survival pathways that are shared by both, tumors and cardiac cells. In this example we treated Ncyte cardiomyocytes with Thapsigargin and performed simultaneous monitoring of contractility and viability in the short and long term.
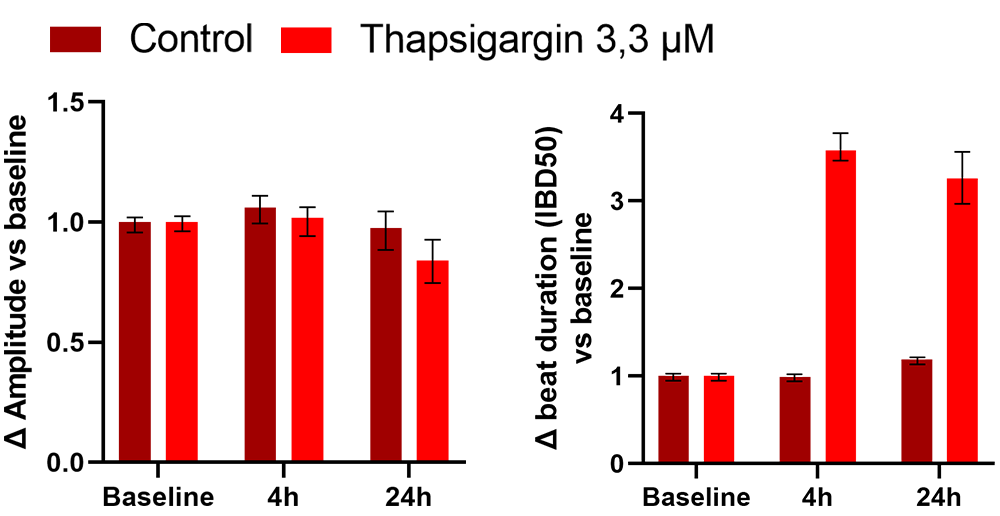 Treatment of human iPSC-cardiomyocytes with Thapsigargin 3,3uM decreases amplitude and triplicates beat duration as measured with impedance analysis.
Treatment of human iPSC-cardiomyocytes with Thapsigargin 3,3uM decreases amplitude and triplicates beat duration as measured with impedance analysis.
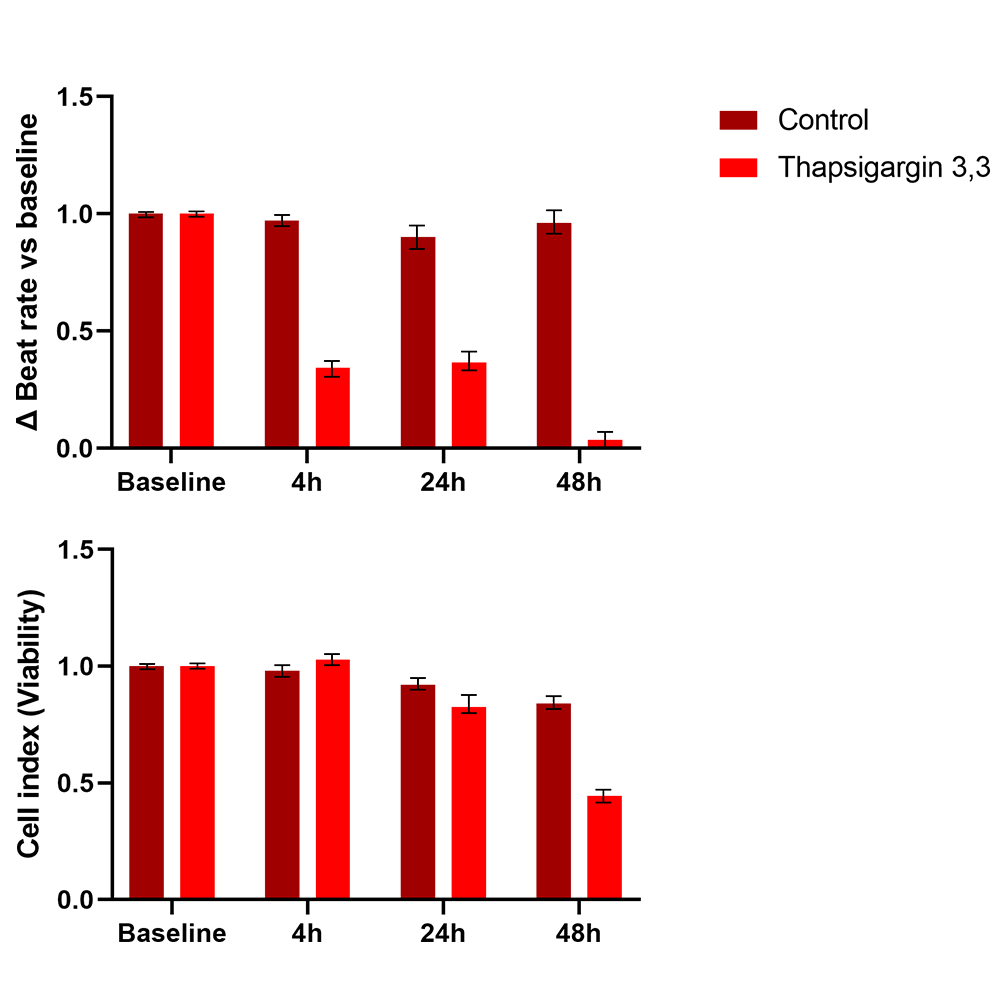 Treatment of human iPSC-cardiomyocytes with Thapsigargin 3,3uM induced a beat rate reduction higher than 50% after 4 and 24 hours and decreased cell viability after 48h
Treatment of human iPSC-cardiomyocytes with Thapsigargin 3,3uM induced a beat rate reduction higher than 50% after 4 and 24 hours and decreased cell viability after 48h
Our work centers on a simple yet powerful premise:
When we combine deep iPSC knowledge, broad assay capabilities and a demonstrated ability to integrate the biology of human diseases into preclinical research, we can help drug developers make critical decisions earlier and with more confidence.
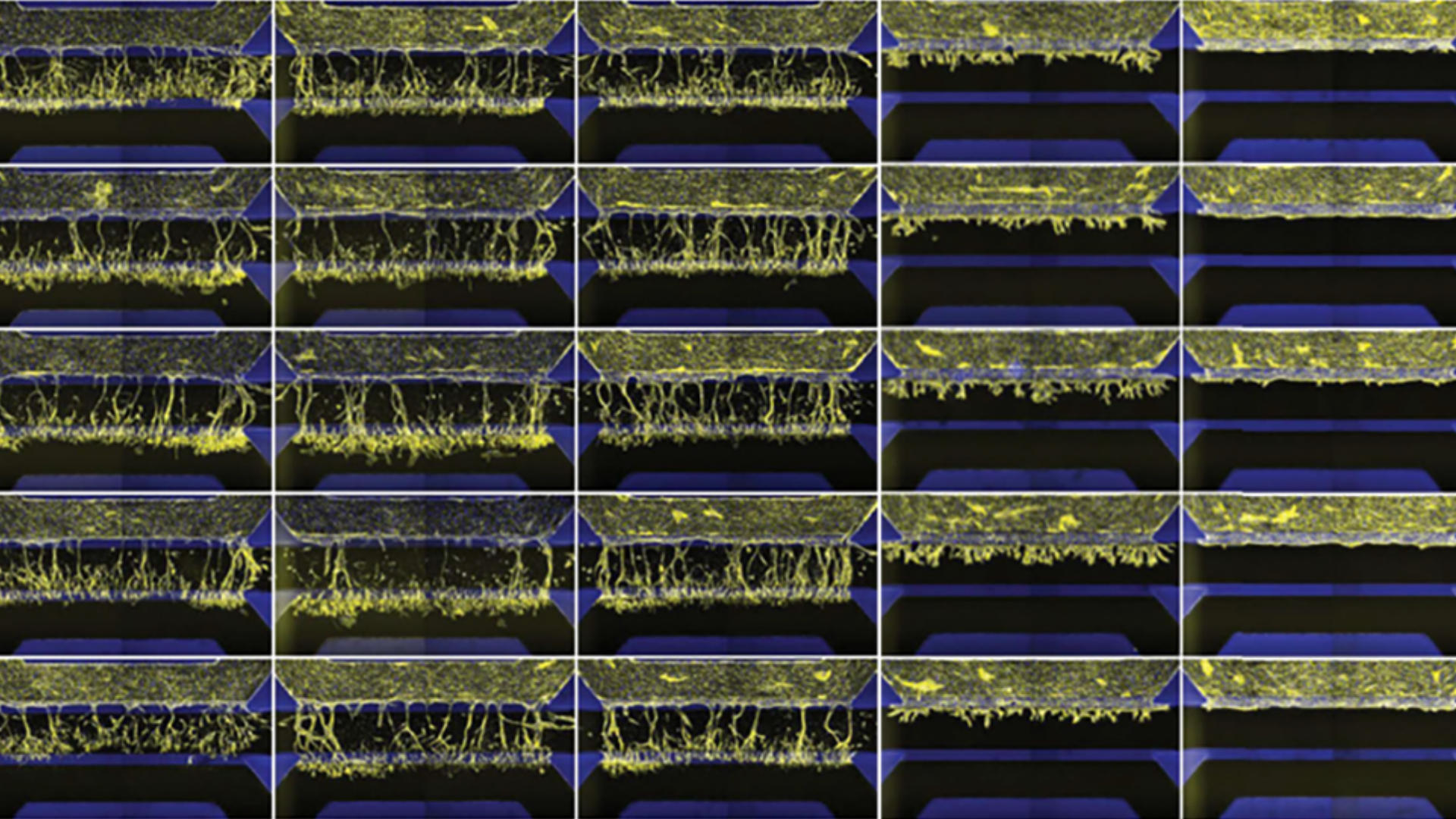
Study compound-induced effects on blood vessel formation
Assay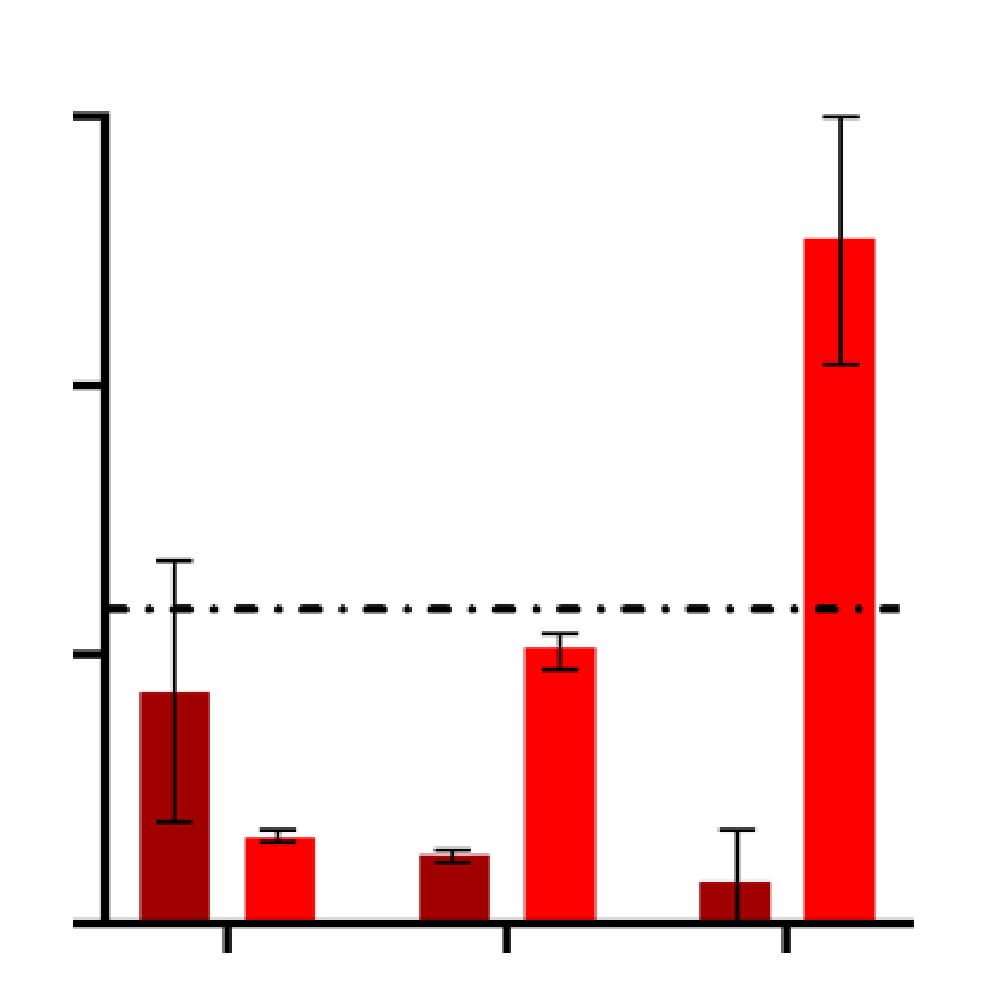
Evaluate and quantify the levels of clinically relevant biomarkers
Assay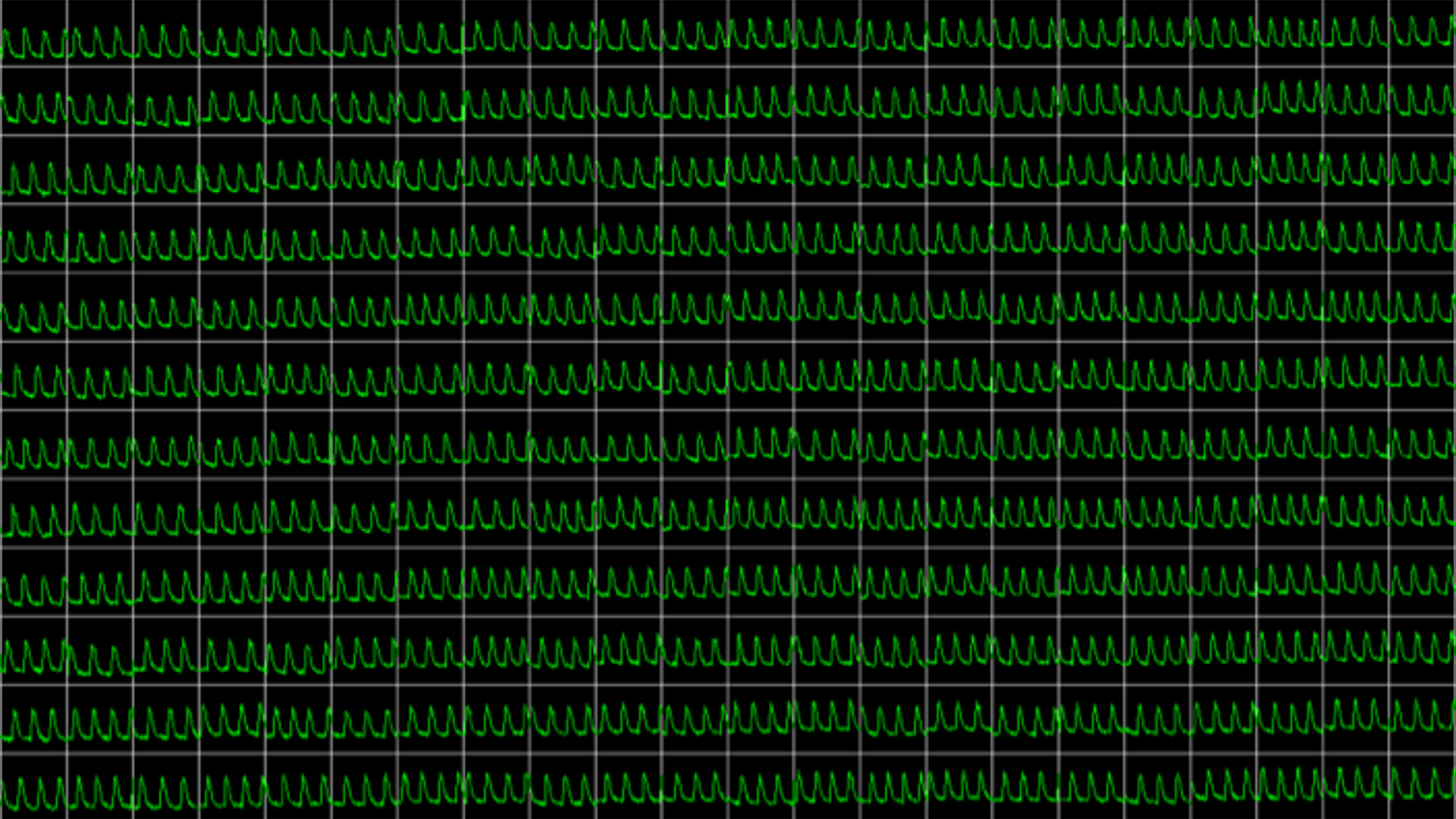
Obtain real-time recordings of intracellular calcium fluctuations
Assay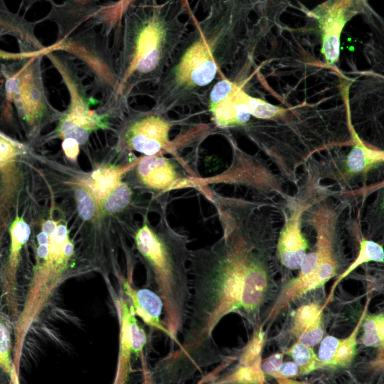
Get in-depth and unbiased insights into the effects of therapeutic candidates on cells
Assay
Study drug-induced effects on contractility of cardiac or skeletal muscle cells
Assay
Determine the electrophysiological effects of your therapeutic candidates
Assay
Evaluate endothelial permeability in short and long term
Assay
Study drug-induced phenotypic changes in the cell model of your interest
Assay
Assess functional toxicity across multiple human iPSC-derived tissues in a single integrated workflow to support safer IO development decisions.
Assay
Obtain precise information on compounds' impact on metabolic processes